Organoids emerge as powerful tools for disease modeling and drug discovery
That's because organoids can mimic many of the anatomical and functional features of a wide variety of human tissues, and, for some tissue types, can be created more efficiently than other models such as cell lines or animal models.
That's because organoids can mimic many of the anatomical and functional features of a wide variety of human tissues, and, for some tissue types, can be created more efficiently than other models such as cell lines or animal models. Their three-dimensional organization can capture much of the complexity of organ structures and cellular relationships, which may be more relevant to real patient tissues. And organoids derived from patients may provide individualized models of disease that are a step closer to precision medicine.
Researchers at the Broad Institute of MIT and Harvard and beyond have spent years developing and refining methods for growing organoids of various kinds. In the last few years, those methods have advanced enough to allow researchers to derive organoids in a reliable, reproducible manner, making them a viable research tool that offers many advantages.
Organoids are far more accessible for laboratory tests than whole organs or lab animals. They can be genetically engineered or treated with chemicals to explore the workings of specific genes, proteins or pathways. They also lend themselves well to high-resolution imaging and single-cell genomics or transcriptomics, revealing a wide variety of scientific insights.
Here are snapshots of three research groups at the Broad deriving and using different types of organoids to look for new drug targets and find mechanisms of disease.
Brain organoids: a window into neurodevelopmental disease
As a developmental neurobiologist, institute member Paola Arlotta wants to understand the development and function of the human brain, especially the cerebral cortex, which controls our most complex—and arguably our most human—behaviors. But studying rodents and neuronal cell lines will only get her so far.
"The human brain has changed through evolution compared to even our closest relatives," said Arlotta, who is also chair of Harvard University's Department of Stem Cell and Regenerative Biology and an associate member of the Stanley Center for Psychiatric Research at Broad. "Therefore, there is only so much that we can learn about how our own brain is made by studying the brains of other species."
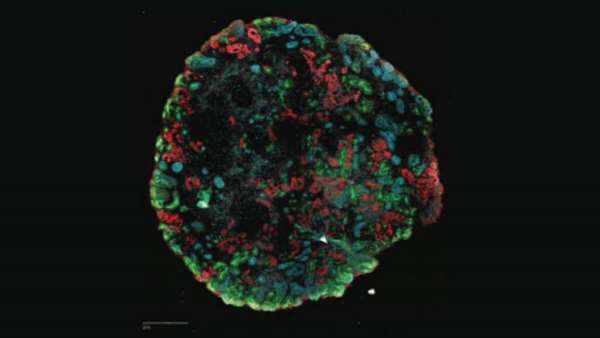
Human brain organoids can fill some of that gap. These raisin-sized organoids lack blood vessels and recognizable brain structures or layers, but at an ultrastructural level, the tissue looks nearly indistinguishable from that of a real brain, with most key cell types, connections between neurons, and even active circuitry.
What's more, if brain organoids are grown from cells with gene variants that have been linked to complex psychiatric or neurodevelopmental disorders such as schizophrenia and autism, they may recapitulate the effects of these genetic changes in a more informative way than nonhuman models. "We never had the right experimental system to ask what mutations in disease risk genes do in the human brain, and that's what the organoids really gave us," said Arlotta.
She and her lab have devoted several years to improving organoid protocols and developing methods to grow the tissues for longer periods—months to years—to better represent later stages of brain maturity. In a 2019 Nature paper, Arlotta and her colleagues showed that they could generate long-growing organoids that make the full gamut of cortical cell types, even when produced from different stem cells and under different growth conditions.
"Most importantly, the organoids could make cortical cells reproducibly, making the same set of cells in each organoid every time," she said.
Reliable methods of generating brain organoids have allowed Arlotta's group to now use them to unpack the biological complexity of neurodevelopmental disorders, including autism. Autism is most often associated with a complex genetic background in which many different gene variants likely contribute to the disorder. But in rare cases, the development of features linked to autism may be strongly associated with one or a few specific mutations. Arlotta's group is generating organoids that carry such autism-associated mutations, and then comparing them with those grown from the same cell lines but without the mutations.
"This type of work is foundational" to understanding how genetic changes affect the function of brain circuits over time, Arlotta noted. Researchers can also compare brain organoids containing the same mutation in different genetic backgrounds, which Arlotta believes will have strong influences on the manifestation of the disorder. Someday, perhaps, brain organoids generated from cells from individuals with autism could yield further clarity about the breadth and diversity of the underlying genetics.
Reference:https://www.nature.com/articles/s41586-019-1289-x



ارسال به دوستان